Southwestern blotting studies that detect DNA-binding proteins. Northwestern blotting studies that detect interactions between RNA and proteins. To study genetic changes in DNA it can be used for homology-based cloning too. The membrane used is a nitrocellulose membrane. The membrane used is an amino benzyloxymethyl filter paper membrane. 
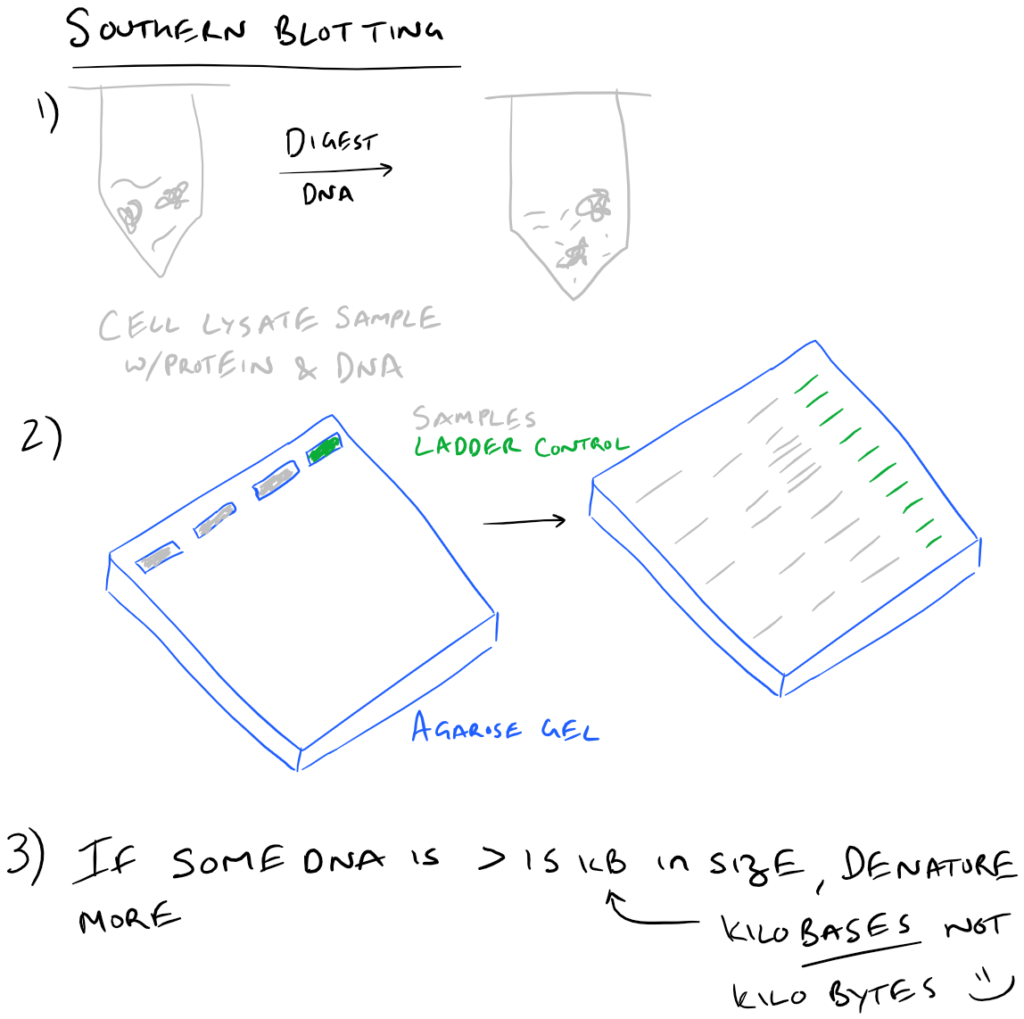
It detects changes in specific DNA sequences. It detects presence of specific RNA sequences. The name is a misnomer―an eponymous derivative of Southern blotting. It was developed by Sir Edwin Mellor Southern in 1975. It was developed by James Alwine, David Kemp, and George Stark in 1977. However, there are some cases where one has to use radioactive labeling. Nowadays, fluorescent dyes are preferred over radioactive labeling to prevent undue exposure to radiation. If a chromogenic dye is used, the results can be seen on the membrane itself. In case of radioactive labeling or the use of a fluorescent dye, an X-ray film is exposed to the membrane for a few seconds and developed. ✦ Depending on the type of secondary probe used, the membrane is visualized accordingly. The membrane is washed gently to eliminate the excess probes, and then it undergoes secondary hybridization, where the target probe is labeled with a radioactive element, a fluorescent dye, or even a chromogenic dye. The binding of the probe to the target molecule is called primary hybridization. The membrane is then treated with specific target probes that bind to the transferred bands. ✦ Once the blocking is done, the membrane is washed gently to remove traces of the blocking solution. This ensures that the transfer is made permanent, and the unbound surface of the membrane is blocked so as to prevent binding of unwanted molecules and the possibility of a hampered result. The membrane with the transferred bands is then sealed by baking in an oven or by treating it with a blocking solution. The bands are then transferred onto a carrier membrane by the principle of either capillary action or directed electric current. ✦ Initially, the purified sample is loaded onto an agarose gel and electrophoresed, causing the sample to separate into bands. The intensity of the resultant dye or fluorescence will indicate the specificity of the binding along with other characteristics such as copy number, gene expression, etc. This change in binding affinity will be visualized upon secondary hybridization with a labeled probe. Any change in the sample sequences will lead to the probes not binding or binding non-specifically. PrincipleĪll blotting techniques are based on the same principle of specific base pairing of the probe sequences to the sequences immobilized on the membrane. The purpose of each technique may be different, but all share the same principle and methodology, with a few minor deviations and modifications. Southern blotting is used in case of DNA, Northern blotting in case of RNA, Western blotting in case of proteins, and Eastern blotting in case of post-translational modifications of proteins. In molecular biology and genetics, various blotting techniques are employed to detect and study changing levels of proteins, DNA, or RNA, and also to study the interactions occurring between them. Subsequent blotting techniques (Northern blotting, Western blotting, etc.), which are based on the same principle, are named eponymously. Southern, a British biologist who developed this technique.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
